SensiLase PAD-IQ
Skin Perfusion Pressure Testing - The Intelligent Vascular Test. Fully automated, quantitative evaluation of microcirculatory perfusion for wound healing prediction and PAD detection.
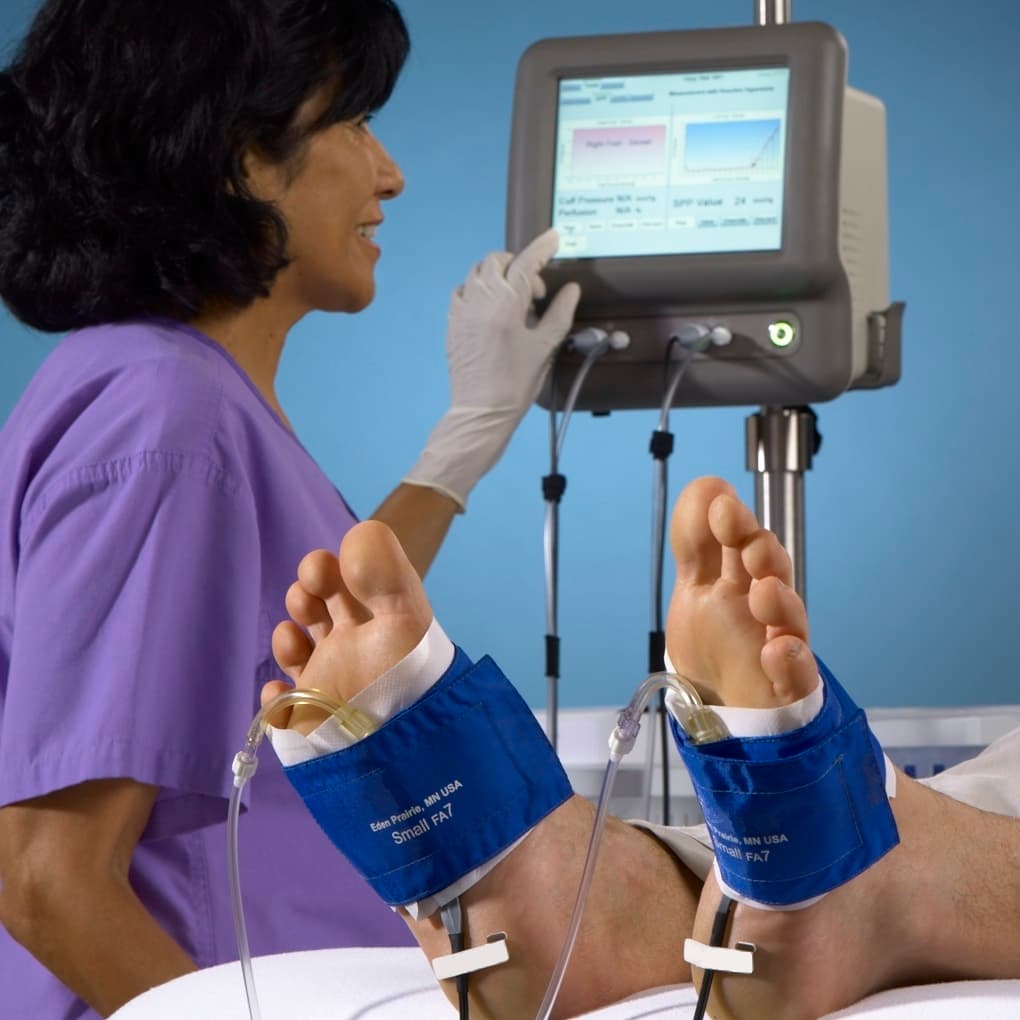
Skin Perfusion Pressure (SPP) is a noninvasive diagnostic test that provides a fully automated and quantitative evaluation of microcirculatory perfusion in the skin. SPP measures blood flow at a depth of 1.5 to 2.0 mm beneath the skin using a laser Doppler sensor combined with a pressure cuff to evaluate reactive hyperemia.
The SensiLase PAD-IQ system delivers dependable, reproducible measurement of tissue perfusion of the extremities, particularly in patients who have combined peripheral arterial disease (PAD) and diabetes.
Key Capabilities:
- Skin Perfusion Pressure - Microcirculation (Reactive Hyperemia)
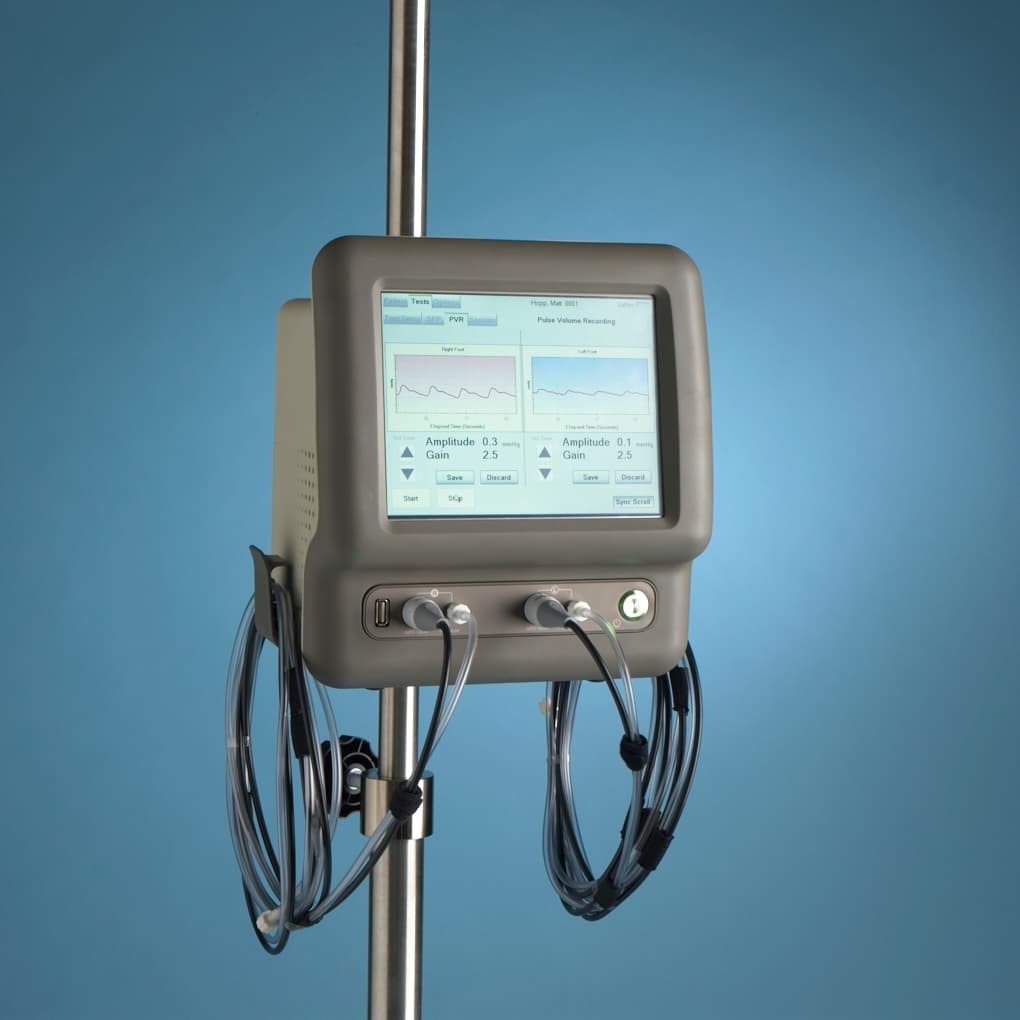
- Pulse Volume Recording (PVR)
- Ankle Brachial Index (ABI)
- Simultaneous Bilateral Testing
- Wound Image Surface Area Calculation
- Angiosome-Specific Testing
SPP measurements provide objective guidance for wound healing potential and treatment planning:
| SPP Value | Classification | Clinical Implication |
|---|---|---|
| > 50 mmHg | Adequate Perfusion | High wound healing potential |
| 40-50 mmHg | Mild Disease | Good healing potential with compliance |
| 30-40 mmHg | Moderate Disease | May require intervention |
| < 30 mmHg | Severe Disease (CLI) | Low healing potential without intervention |
When SPP is ≥ 35mmHg, surgical debridement of necrotic tissue is feasible without peripheral arterial reconstruction.
Works When Other Tests Cannot
SPP measurements are unaffected by conditions that limit other diagnostic tests:
- Calcified arteries - Common in diabetic patients
- Edema - Does not affect accuracy
- Callused skin - Can test plantar foot
- Inaudible pulses - No Doppler signal required
- Anemia/Hypoxia - Measures pressure, not O2
Operational Benefits
- No calibration required
- No contact gel or solution needed
- No heated electrodes
- Minimal training requirements
- Touch screen operation - no mouse/keyboard
- Portable: table, IV pole, or bed mount
- Telemedicine and EHR compatible
- ✓Touch Screen Interface - Complete operation without mouse or keyboard
- ✓Simultaneous Bilateral Testing - Test both limbs at once
- ✓Fully Automated - Consistent, reproducible results
- ✓Wound Imaging - Calculate surface area with SmartGuide
- ✓CPT/ICD Codes - Automatic population for billing
- ✓Remote Interpretation - Telemedicine compatible
- ✓EMR Integration - Encrypted data transmission
Primary Indications
- Wound healing prediction
- Amputation level determination
- Critical Limb Ischemia (CLI) assessment
- PAD detection and severity assessment
- Pre/post revascularization evaluation
- Diabetic foot assessment
Target Patient Populations
- Diabetic patients with PAD
- Patients with chronic wounds
- Hemodialysis patients
- Patients with calcified arteries
- Patients requiring amputation planning
- Post-endovascular intervention monitoring